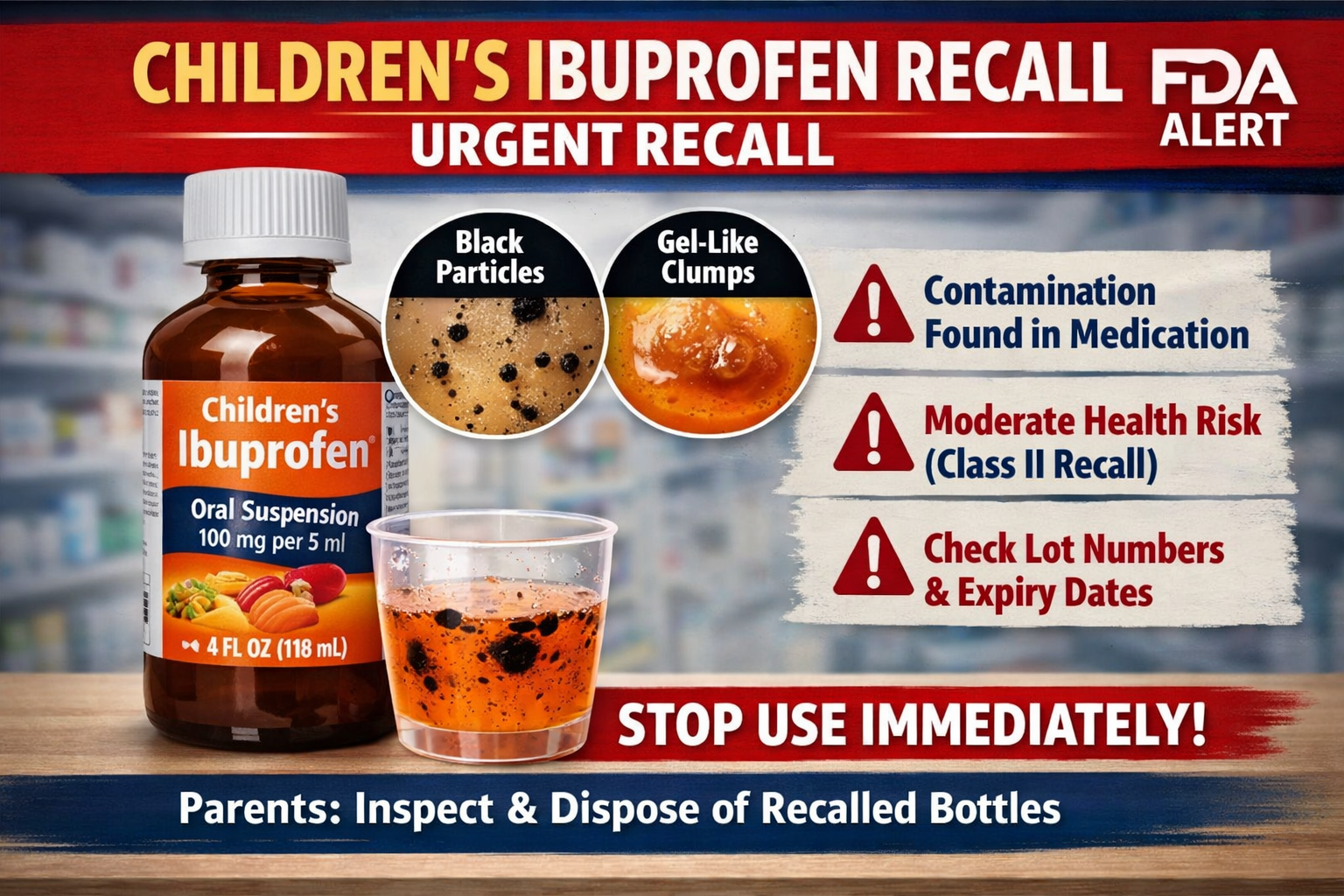
The U.S. Food and Drug Administration (FDA) has issued a recall of approximately 90,000 bottles of children’s ibuprofen nationwide, casting serious doubts on the safety of a popular pediatric drug. Parents and healthcare providers have a strong faith in the use of ibuprofen as an antipyretic medication against fever, inflammation, and mild pain in children. Due to the prevalence of this medication and the ease with which it can be obtained over the counter, any facility related to this drug turns out to be a major subject of health concern among the masses. The recall emphasizes that the quality control of the pharmaceuticals production process should be very strict and it should be continuously monitored to guarantee the safety of the consumers.
Reason Behind the Recall
Several reports of contamination of the product caused the recall. The consumers found foreign substances in the liquid medication that consisted of black particles and gel-like masses. This is a disturbing finding, especially in a drug that is supposed to treat children, which means that there are violations of the traditional rules of manufacturing and safety. The availability of these materials creates the possibility of problems with the production processes, like inefficient filtration, contamination during packaging, or poor storage conditions. Although the actual source cause has not been extensively reported, the identification of visible impurities is enough to recall them instantly to avoid any potential damage.
FDA Level of Classification and Risk
The FDA classified this recall as a Class II recall, which is a moderate risk category. This grouping implies that the product can produce short-term or reversible adverse health effects that can be produced by other factors, but the risk of extreme or fatal effects is not very high. Although the Class II recall is a less serious problem than Class I, it conveys that the product is not safe to ingest. The classification assists the health care providers and the consumers in knowing the degree of the situation and taking appropriate action.
Product Information of the Affected Product
Their product that was later recalled was the Children’s Ibuprofen Oral Suspension, in most cases it is taken in liquid doses to facilitate easy administration to a young child. It is available in a 4-ounce bottle with a 100 mg per 5-ml dose. Certain lot numbers have been determined to be part of the recall, and the products have an expiry date that goes to 2027. Due to this prolonged shelf life, it is likely that a lot of households still have these bottles at home, and it makes the issue of publicity even more critical. It is recommended that the parents and caregivers also scrutinize the packaging information to find out whether their product falls under the recall or not.
Influence on Consumers and Families
The families are directly affected by the recall, and in particular those with young children who often use ibuprofen to treat fever and pain. It might also cause anxiety among parents since they might be worried as to whether their child has swallowed a poisoned product. Although there are no serious health cases reported, the fact that there is a probability of exposure to foreign substances is enough to raise concern. Moreover, the recall might interfere with the regular treatment, leaving families to find alternative drugs or consult healthcare professionals. This may be especially tricky, especially when the children are more susceptible to sickness like seasonal flu attacks.
Wider Problems within Pharmaceutical Supplies
This event also demonstrates larger issues of the pharmaceutical business, and in particular, delivering quality throughout the global supply chain. A large number of drugs are produced in factories in other countries, and hence, regulatory monitoring may become more complicated. Inasmuch as agencies such as the FDA do the inspection and setting of standards, the levels and global nature of production provide room for slack. The generic pharmaceutical market also faces cost pressures that may affect the manufacturing practices, in some cases resulting in a decrease in the investment in quality control systems. Consequently, to guarantee the safety of drugs that are massively spread, close attention, better monitoring systems, and enhanced cooperation between the manufacturers and the regulators are necessary.
Significance of Safety and Quality Standards
Quality control is also another important element of producing pharmaceuticals, particularly those which are supposed to be used by children. All tests should be strictly followed before any drug is released into the market, including contamination tests, consistency tests and stability tests. The inability of these processes can result in recalls, which harm both consumer safety and the reputation of the manufacturers, as well as decrease the trust of people in healthcare products. Such a recall is an educational experience that pharmaceutical companies cannot afford to be any less vigilant about upholding high standards, as they are the central role in such firms.
What Consumers Should Do
The consumers are encouraged to ensure that they act without delay in the event that they have bought children’s ibuprofen. To begin with, they can examine the product label that contains the lot numbers and the expiry dates to tell whether the product is a recall or not. In the event that the product corresponds with the details that have been affected, there should be no cases of using the product. Parents are expected to dispose of the medication in a safe way or by adhering to the instructions on its reuse given by the retailers or manufacturers. In case a child has taken the product already and exhibits any abnormal behavior, one should consult the doctor immediately. Moreover, this could be followed by reporting of any adverse effects in order to help the authorities monitor the situation and act further in case of necessity.