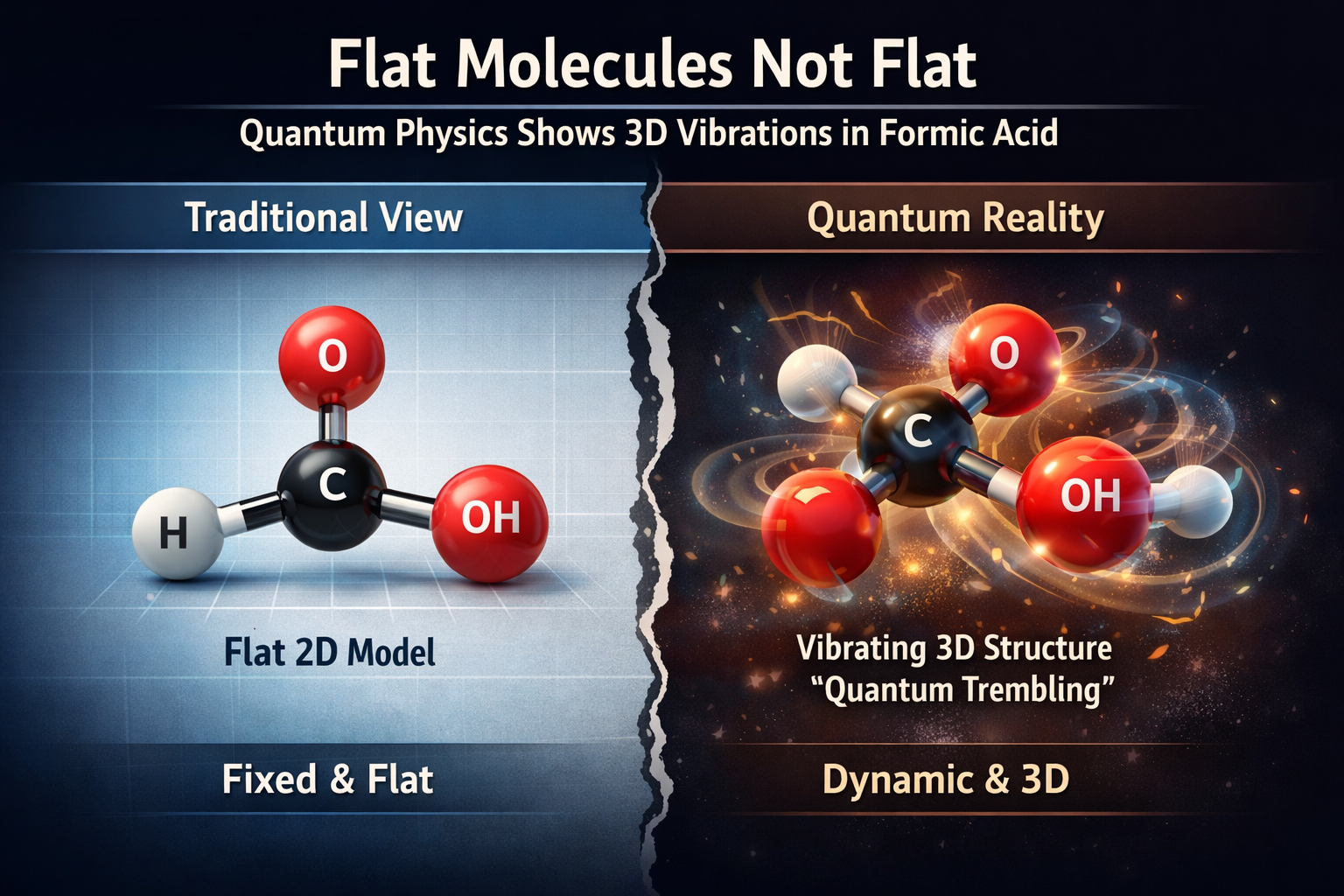
In an intriguing twist that bridges classical chemistry and quantum mechanics, a new study reveals that molecules we’ve long thought were perfectly flat—like formic acid—are never truly flat at all. While textbook diagrams depict planar molecules as lying cleanly in two dimensions, researchers using cutting-edge experimental techniques have shown that quantum mechanical effects make these molecules gently tremble into the third dimension, even at the smallest scales. This finding challenges simplistic geometric assumptions and highlights the dynamic, probabilistic nature of molecular structure in the quantum world.
The Traditional View vs. Quantum Reality
In high school chemistry and countless scientific illustrations, certain molecules—such as benzene or formic acid (HCOOH)—are represented as flat, two-dimensional arrangements of atoms. These depictions help students and scientists alike visualize structures and predict reactivity. In these models, atoms are shown as dots or spheres connected by fixed lines representing bonds.
However, researchers note that such visualizations are approximations. At a fundamental level, quantum mechanics governs how particles behave. In the quantum realm, atomic nuclei are not fixed points with precise positions; they’re described by wave functions and probabilities, making their locations inherently uncertain. As a result, even a molecule that appears flat on paper exists as a fuzzy, dynamic structure in three-dimensional space.
How Scientists Studied Molecular ‘Flatness’
A team led by physicists at Goethe University in Frankfurt, Germany, sought to test just how “flat” a planar molecule really is. They homed in on formic acid—a simple organic acid used in various industrial and biological contexts. In its most basic geometric representation, formic acid is strictly planar, with all atoms lying in a single plane.
To investigate, the researchers used high-energy X-ray beams from a synchrotron particle accelerator, specifically the PETRA III source at DESY, to bombard formic acid molecules. This intense radiation ionized the molecules, causing them to undergo a Coulomb explosion—a rapid dissociation that ejects electrons and scatters nuclei. By analyzing the directions and energies of the many electrons and fragments produced, researchers could mathematically reconstruct the original structure of the molecule in exquisite detail on ultra-short timescales.
The result? What was assumed to be flat exhibited small but consistent vibrations, particularly in its hydrogen atoms. These vibrations are driven by quantum mechanical effects and mean that formic acid spends almost all of its time in slightly non-planar conformations—effectively three-dimensional shapes. The team dubbed this phenomenon “quantum trembling.”
What Quantum Trembling Means
This quantum trembling implies that formic acid—and potentially many other so-called flat molecules—doesn’t actually remain in a perfectly flat configuration at the quantum level. Rather, atoms are constantly fluctuating due to inherent quantum uncertainties. Even though average geometrical parameters (like bond angles) may match textbook values, the instantaneous positions of atoms are dynamic, not fixed.
One especially striking aspect is how such motion can influence molecular handedness (chirality). The vibrations can nudge the molecule between left-slanted and right-slanted forms, showing that what might be considered a static, symmetrical structure actually oscillates between different effective geometries.
This doesn’t mean classical chemistry is wrong. Molecular modeling and predictions based on flat geometries remain useful, especially in reaction mechanisms and substance design. But it reveals a deeper nuance: molecular shapes are not rigid assemblies but ever-moving quantum entities.
Why This Matters in Chemistry and Physics
Dynamic Reality vs. Static Models
The research underscores an important distinction: models are simplifications. Chemists use them because they work well for predicting reactions and explaining properties. But at the quantum level, nature does not adhere to our simplified diagrams. Electrons and nuclei exist in probabilistic states, and their motion contributes to properties that may only be apparent through precise quantum mechanical understanding.
Impact on Chemical Understanding
Quantum trembling could influence how scientists think about molecular reactivity, stereochemistry, and even the behavior of complex biologically relevant molecules. For example, molecular chirality—a key factor in drug design and biochemical interactions—might depend not just on static shape but on the dynamic fluctuations of atoms that can tilt a structure in subtle ways.
Broader Quantum Insights
This finding also resonates with broader principles in quantum physics. Particles exhibit wave-particle duality, meaning they behave both as discrete particles and as waves that spread through space—with positions best described by probabilities rather than certainties. (This parallels matter waves in quantum mechanics, where particles like electrons have associated wave functions that describe where they’re likely to be found.)
The fact that even low-energy molecules like formic acid exhibit such intrinsic motion highlights the ever-present influence of quantum behavior—even in systems traditionally investigated in classical contexts. That molecules wobble in three dimensions simply because of quantum effects echoes how quantum uncertainty and zero-point energy prevent any particle from being completely at rest, even at absolute zero.
How This Research Fits in Scientific Progress
Quantum chemistry and molecular physics have long acknowledged that atoms and electrons behave according to quantum rules. Computational techniques—such as molecular orbital theory and quantum mechanical simulations—already account for some degree of motion and uncertainty.
But experimental confirmation of quantum trembling in a molecule often assumed to be flat helps bridge theory and reality. It shows that experimental techniques are now sensitive enough to detect subtle quantum effects in molecules, not just infer them through indirect calculations. This represents progress in our ability to visualize and understand the quantum dynamics of matter.
Looking Forward: What’s Next?
This research opens several potential avenues for future exploration:
- Studying other “flat” molecules: Do planar systems like benzene or polycyclic aromatic hydrocarbons exhibit similar quantum trembles? How might that affect their electronic properties or reactivity?
- Biochemical implications: Proteins and large biomolecules have highly complex three-dimensional shapes. Quantum motion might subtly affect folding, function, or interaction profiles.
- Material science: Understanding the intrinsic motion of atoms in solids, surfaces, and interfaces could refine models in nanotechnology and materials engineering.
- Future studies may combine experimental techniques with advanced quantum simulations to map out the full dynamic landscape of molecular geometries, bridging the gap between probabilistic quantum states and observable macroscopic behavior.